As for hydrogen, the weight of 1.6mg at the volume of 17.6ml melts for 1 liter of water (water temperature around 20 degrees Celsius) Generally the number of saturated 1.6pp may be common. However, a unit of mol is more desirable scientifically.
Thus 1.6ppm is 0.8 millimeter of mol/liters.
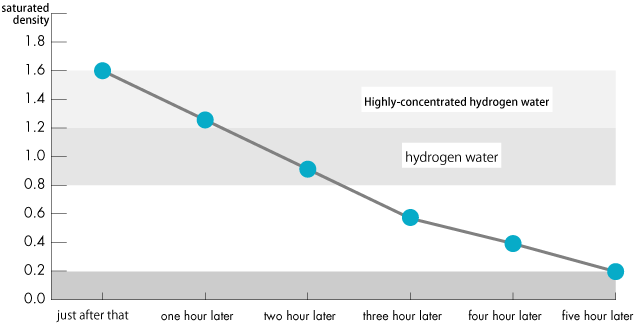
The Amount Change of Molecular Hydrogen after pouring hydrogen water into a cup
Numerical value of hydrogen concentration is a little bit different by the type or the pressure of a container.
The numerical value of the hydrogen residual quantity after pouring H₂ water into a cup is as follows.
Some people think that hydrogen falls out immediately.However, you can take in enough hydrogen even after you put the hydrogen water into a glass at the time of meal and drink it.
In addition please be careful about the products ( hydrogen water, hydrogen water generation machine) declaring“there is no change in a physical quantity of hydrogen by a special technique even if several hours pass.”
There is no significant difference in passing through as characteristic of hydrogen.
Such characteristic (diffusibility and molecular size) that is easy to pass through is effective to eliminate active oxygen.
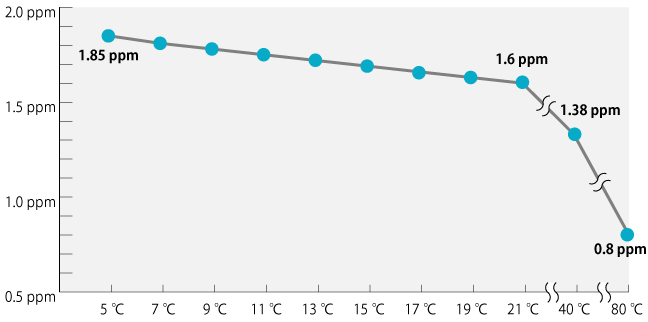
Relations of the hydrogen saturated hydrogen concentration and temperature
Please look at the chart. When water temperature becomes higher, the saturated density lowers.
Generally, the saturated density of hydrogen water is 1.6ppm. Scientifically it is universal that the gas melts a lot when temperature is low.
Temperature does not matter much with molecular hydrogen. Please drink hydrogen water at a temperature for suitable to drink every day.
This line graph is related to the time of production. And thus, density cannot rise even if you cool it in the fridge.
And this density value changes by pressure and impurities.